By Denis Katz, MD
Strategic Frameworks for Clinical, Regulatory, and Commercial Success
Founder, Salience Clinical, LLC
Executive Summary
Musculoskeletal regenerative medicine programs with rigorous biomarker strategies reach market 18-24 months faster than competitors and achieve breakthrough designation rates 2-3x higher than the industry average. Despite this, fewer than 25% of the 40+ programs currently in development have strategically aligned biomarker approaches.
Key Strategic Insights:
- Avoidable Waste: Inadequate biomarker strategies cost an average of $40-80M in trial expenses and 12-18 months in regulatory delays.
- The Multi-Modal Advantage: Integrating molecular, imaging, functional, and digital markers creates a 3-4x stronger evidence package.
- Market Shaping: First movers who validate biomarker-outcome correlations set the regulatory and reimbursement standards for the entire category.
The Board Meeting That Lasted 11 Minutes
Four years of work. $180 million spent. And the Phase III trial had failed.
Not because the science was wrong—the regenerated cartilage looked beautiful on MRI. Not because of safety issues. The trial failed because nobody could prove the regenerated tissue actually helped patients walk better, climb stairs, or reduce pain in meaningful ways.
The company had invested heavily in structural imaging but failed to validate whether “prettier” cartilage correlated with patient function. By the time they discovered the disconnect, they’d burned through their capital.
The Salience Perspective: A properly designed strategy would have integrated MRI findings with functional biomechanics and patient-reported outcomes in Phase I. The cost to de-risk this? Approximately $400K. The cost of ignoring it? $180M.
Self-Assessment: Is Your Program at Risk?
If you answer “No” to any of these and are past preclinical development, you have a strategic gap actively costing you time and capital.
- Prediction: Can your biomarkers predict clinical outcomes 6-12 months before traditional endpoints?
- Validation: Have you validated biomarker-outcome correlations prospectively in your target population?
- The Payer Story: Do your biomarkers connect tissue mechanism to cost offsets, avoided procedures, or functional improvement?
The Multi-Modal Framework
The most successful programs use 3-4 complementary biomarkers to create overlapping evidence chains.
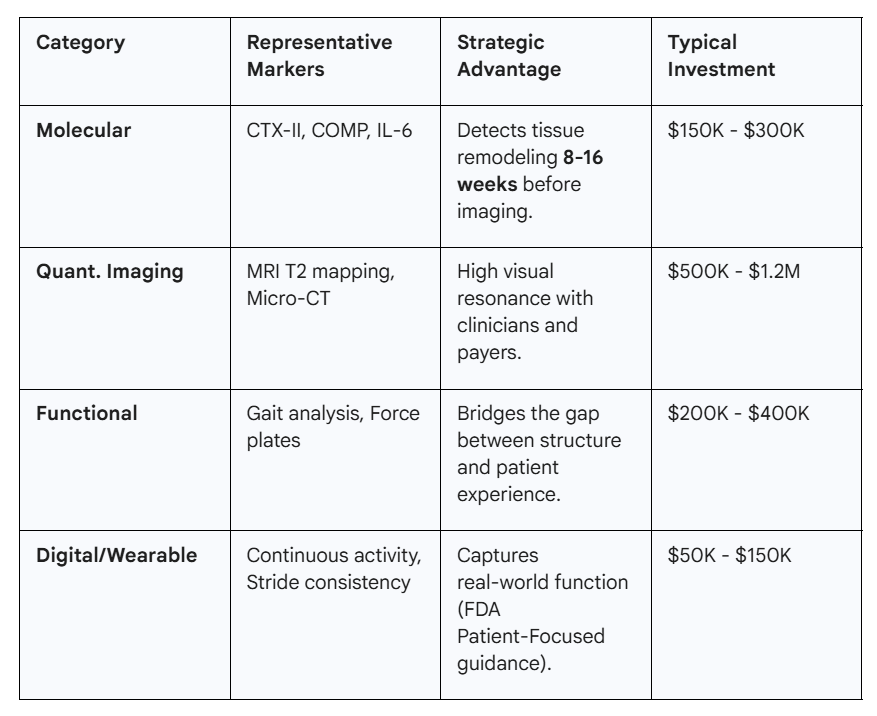
Navigating Regulatory Pathways
FDA and EMA offer multiple accelerated pathways, but they are “biomarker-hungry.”
- Breakthrough Therapy Designation (BTD): Requires preliminary clinical evidence of “substantial improvement.” Programs with robust biomarker packages see a 35-40% grant rate vs. the 15% industry average.
- RMAT Designation: Specifically for regenerative therapies. Provides early interaction opportunities with the FDA to align on validation before pivotal investment.
- Accelerated Approval: Success depends on surrogates “reasonably likely to predict clinical benefit,” requiring deep natural history data.
The Validation Framework Regulators Expect
- Level 1: Analytical Validity (Reliability)
- Requirement: Inter-laboratory reproducibility (CV <15%).
- Level 2: Clinical Validity (Relationship to Disease)
- Requirement: Prospective correlation studies in your specific target population.
- Level 3: Clinical Utility (Decision-Making)
- Requirement: Does the biomarker improve outcomes or efficiency vs. not using it?
The Payer Value Tier Framework
Payers don’t care about “beautiful science”; they care about budget impact.
- Tier 1: Basic Coverage: Non-inferiority to standard care. (Parity pricing).
- Tier 2: Preferred Coverage: 18-24 month durability data + responder identification. (15-25% premium).
- Tier 3: Premium Coverage: Early responder prediction (≥80% accuracy) + quantified cost offsets like surgical avoidance. (30-50% premium).
When Your Primary Biomarker Fails: Recovery Playbooks
Scenario: Biomarker Improves, But Clinical Outcomes Don’t
- Immediate Action: Analyze correlation patterns. Do not panic.
- Strategy: Extend follow-up duration. Structural improvements in musculoskeletal tissue often precede functional adaptation by 6-12 months.
- Outcome: Use the “structural-to-functional” lag data to refine Phase III endpoints.
Scenario: Regulators Reject Your Surrogate
- Immediate Action: Seek a Type C meeting focused exclusively on validation requirements.
- Strategy: Pivot to using the biomarker for patient enrichment (selecting those most likely to respond) even if it isn’t the primary endpoint.
Your Next 90 Days: A Strategic Action Plan
- Weeks 1-4: Map current biomarkers against the four strategic functions (Acceleration, Regulatory, Adaptive Design, Payer Value).
- Weeks 5-8: Schedule a Type C or pre-IND meeting with the FDA to align on the validation package.
- Weeks 9-12: Initiate analytical validation and build preliminary health economic models.
The companies defining biomarker standards today—demonstrating what “good” looks like to regulators—will control the market for the next decade.
Work With Salience Clinical
If you are developing musculoskeletal regenerative therapies and need to strengthen your evidence strategy, we offer focused consultations on:
- Biomarker portfolio optimization.
- Regulatory pathway alignment and FDA engagement.
- Payer value proposition development.
